|
11/28/2023 0 Comments Iso 13485 2003 pdf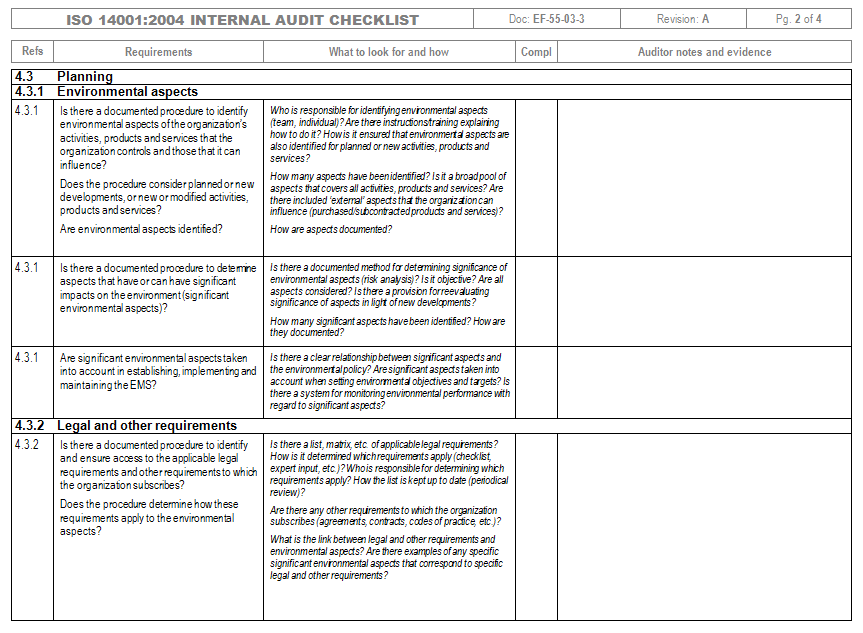 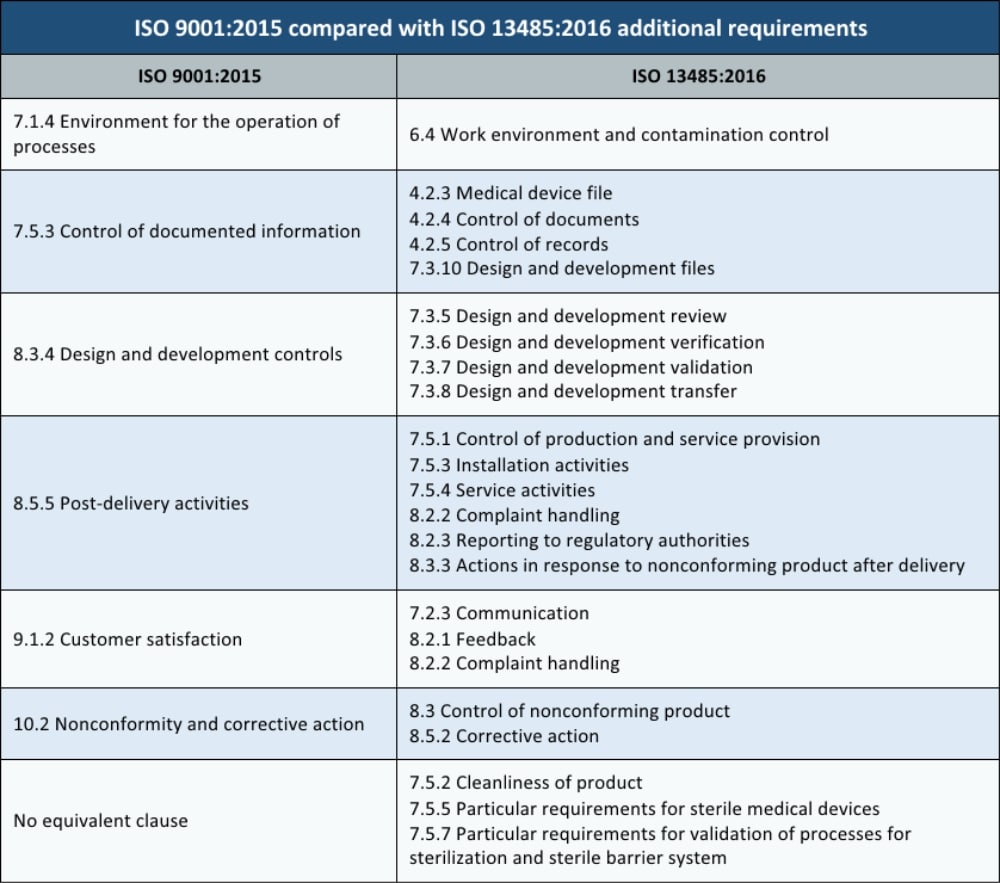
The International Organization for Standardization (ISO) published the updated ISO 13485 medical devices quality management systems standard on March 1, 2016. This white paper is designed to clear up any misunderstandings regarding documents required by this standard, as well as outlining non-mandatory documents that are commonly used. 
Knowing what documents and records are necessary for ISO can be confusing. Checklist of Mandatory Documentation Required by ISO (PDF) White paper. ISO 13485 (medical devices-quality management systems-requirements for regulatory purposes) is an international standard that presents the requirements for a quality management system specific for the realization of medical devices, including software systems with medical purposes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
